TEST KIT
Our easy to use saliva test allows you to measure your deuterium level before and after your deuterium depletion protocol. Also test your water for deuterium levels.

PROVEN BENEFITS
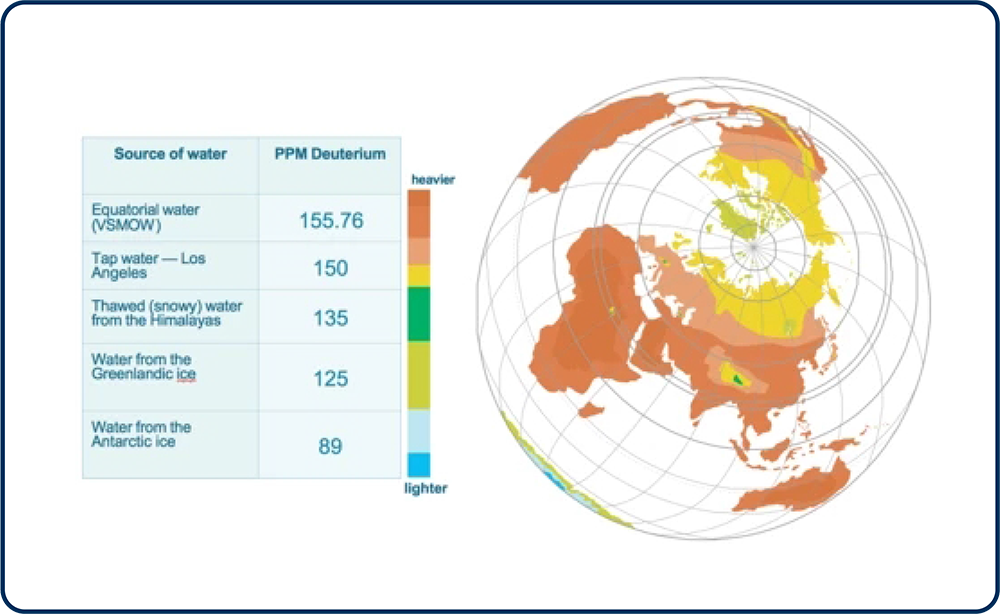
The average person has a deuterium level of 150 ppm and in areas of the earth known for better health, the deuterium level of the water and people is reduced by 15- 20%.

HEALTH AND LONGEVITY
Studies show that reducing deuterium level contributes to a better health and longer lifespan. Deuteriumdepletion.org is a great resource for those researching the new science of deutonomics.
HOW IT AFFECTS YOU
Deuterium and the Body

Did You Know?
Deuterium reduces our ability to make ATP, the main energy currency of the body.

Deuterium & the earth
On Earth, in every liter of regular water there are six drops of heavy water or about one deuterium per every 6600 protium atoms, which is about 150 ppm. Earth's waters are from 89-155 ppm.

Accumulation in the body
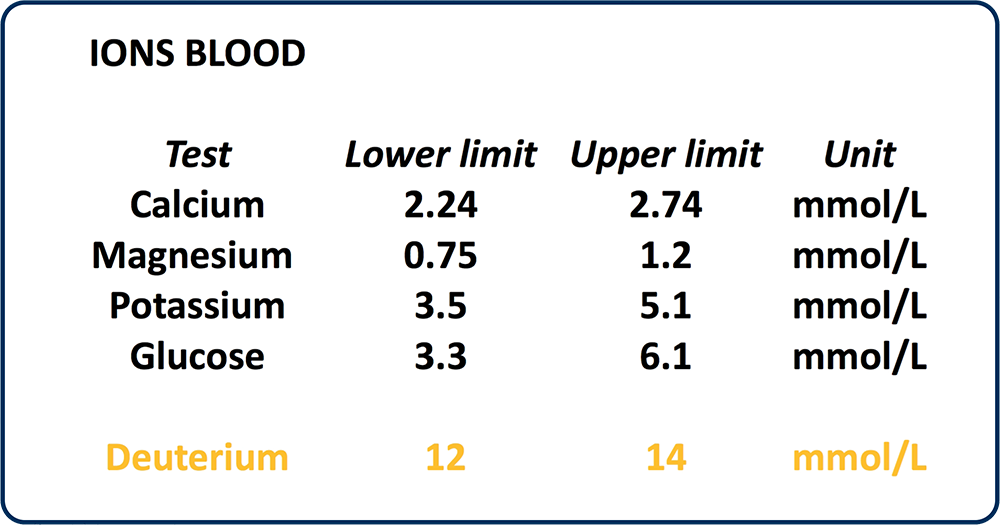
Although deuterium makes up .0125% of all hydrogen, our bodies are continuously burdened. There is 4 times more deuterium in blood plasma than glucose, the most vital nutrients needed for life.

Aging process
The body's try to manage deuterium but accumulation over the years makes it difficult to manage until we become overloaded. Deuterium interference is being researched as a main cause of aging.
The amount of deuterium in the body is an important biomarker of health and
longevity, so if you are currently on a deuterium depletion protocol or are just
getting started it is important to know your total body burden of deuterium.
This easy to use saliva test can be done at home, takes just a few minutes and
delivers fast and accurate results.
Simply order a test kit, follow the instructions, mail back the sample in the prepaid envelope and in 10-14 days you will receive your results by email. All
results are secure and confidential.
Testing is done in a laboratory using the LGR Triple Isotopic Analyzer
Analyzer, providing best in class accuracy for measurement of deuterium.

Did You Know?
You can also use this test kit to measure deuterium levels of water and other liquids.

Step one
Order your test kit. It takes about 3 days to receive it.
Step two
Follow the instructions to collect your saliva. This takes about 5 minutes.
Step three
Mail back in the envelope provided and receive your results in about a week.
ORDER YOUR KIT
DEUTERIUM 101
Facts and questions
WHAT IS DEUTERIUM 'D'?
Deuterium is one of the three forms (called isotopes) of the first element, hydrogen. (get from standard definitions). The first hydrogen isotope is named ‘protium’ (the most abundant by far and the lightest), consisting of a proton and an electron. Protium is commonly expressed by the symbol ‘1H’. The second isotope is named ‘deuterium’ (much less abundant than protium but twice as heavy due to the addition of a neutron). Deuterium is commonly expressed by the symbol ‘D’ or ‘ 2H’. The third isotope is named ‘tritium’ (almost non-existent on Earth and three times as heavy as protium due to the addition of two neutrons). Tritium is a radioactive and luminous gas. The most important fact to know is that in nature, both protium and deuterium combine with oxygen to form water molecules.
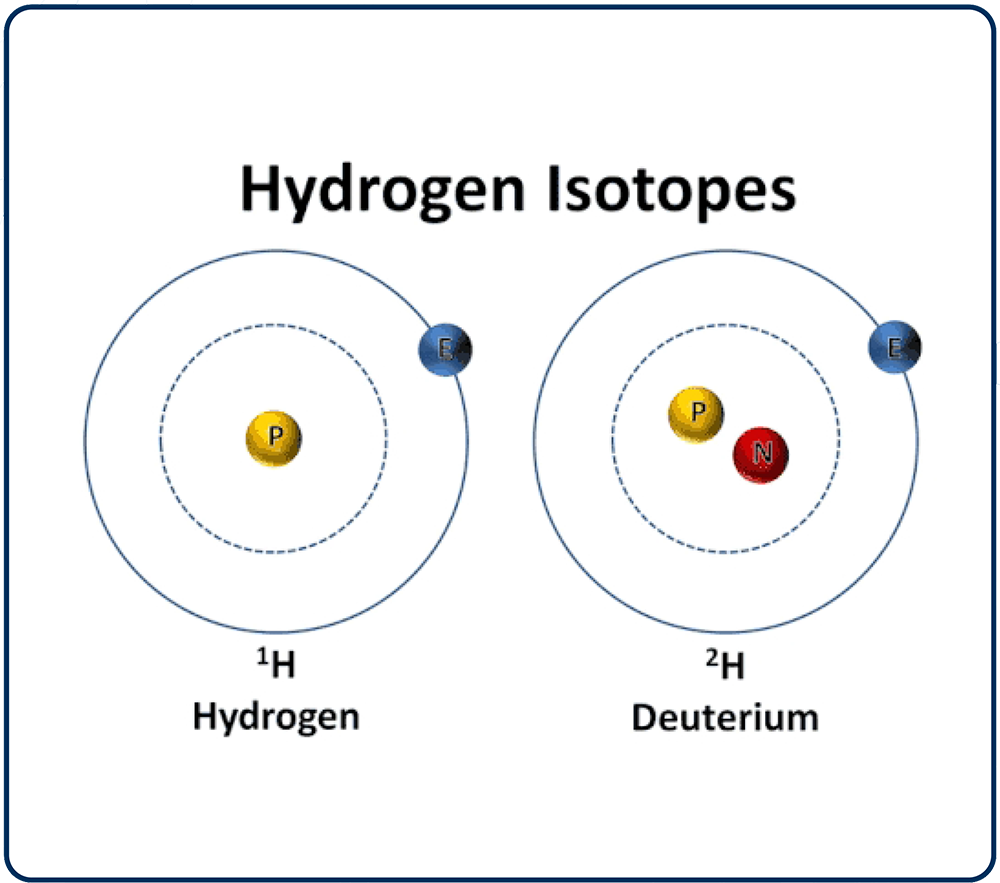
PREVALENCE OF HYDROGEN ISOTOPES ON EARTH
Protium - 99.98% (by number) Deuterium - 0.0156% (by number) On our planet, for every 1,000,000 atoms of Hydrogen there are about 150 atoms of deuterium, (150 ppm).
PREVALENCE OF HYDROGEN ISOTOPES OF DEUTERIUM IN BLOOD
Even though there is only 1 deuterium for every 6600 hydrogen in water, there is 4 times more deuterium in human blood than the most important life giving elements and nutrients!
HOW ACCURATE IS THE DEUTERIUM TEST?
Our testing lab uses the Los Gatos research deuterium analyzer. This instrument uses proprietary laser absorption technology called Off-Axis Integrated Cavity Output Spectroscopy *OAICOS+ that is highly effective and makes the analysis of a large number of samples possible with great accuracy, efficiency and affordability.
Rapid Commun. Mass Spectrom. 2016, 30, 415ò422 *wileyonlinelibrary.com+ DOI: 10.1002/rcm.7459
Measurement of extremely 2 H-enriched water samples by laser spectrometry: application to batch electrolytic concentration of environmental tritium samples L. I.
Wassenaar*, B. Kumar, C. Douence, D. L. Belachew and P. K. Aggarwal International Atomic Energy Agency, Vienna International Center, A-1400, Vienna, Austria